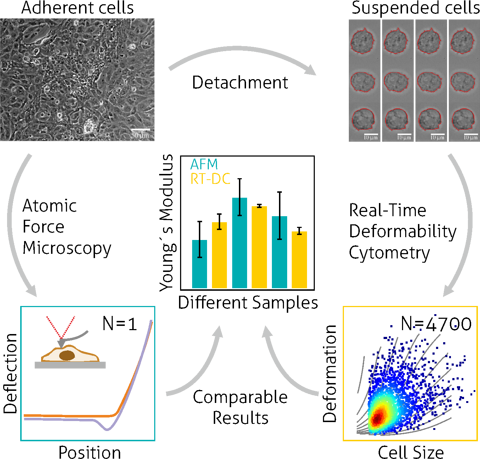
The mechanical phenotype of cardiomyocytes derived from human induced pluripotent stem cells (hiPSC-CMs) provides essential insight into their maturation state, cytoskeletal integrity, and functional capacity in regenerative medicine and translational research. High-throughput, label-free cell analysis enables non-invasive quantification of intrinsic mechanical properties such as deformation, stiffness, and viscoelastic response without fluorescent markers or staining, which complements conventional molecular and electrophysiological readouts.
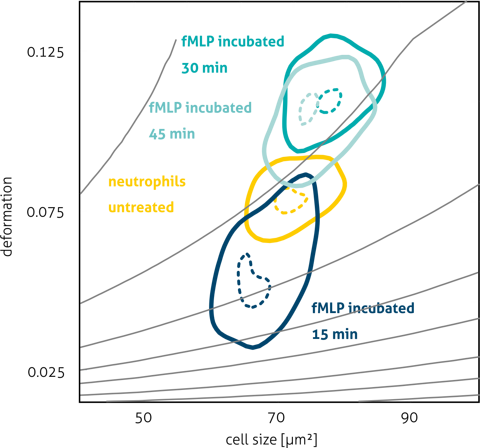
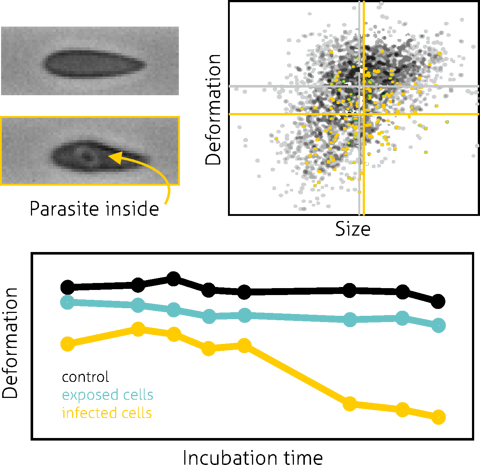
Real-time deformability cytometry (RT-DC), a microfluidic deformability cytometry platform, is particularly well suited for cardiomyocytes because it can probe thousands of suspended cells per second, enabling robust assessment of population heterogeneity and mechanical maturation without laborious sample prep. In hiPSC-CMs, mechanical phenotyping captures changes in cytoskeletal dynamics and cellular elasticity that track with differentiation and structural organization.
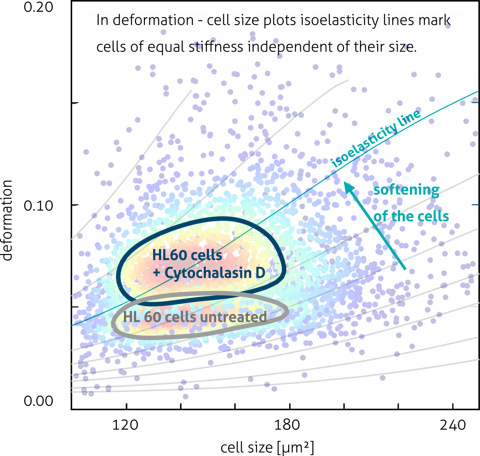
In a seminal study by Pires et al., RT-DC was used to determine the mechanical properties of hiPSC-derived cardiomyocytes in suspension, reporting a Young’s modulus in the range expected for early-stage cardiac cells and revealing distinct regimes of cellular elasticity linked to cytoskeletal actin organization. These measurements demonstrate the feasibility of using label-free mechanical phenotyping to monitor cardiomyocyte maturation, cytoskeletal integrity, and response to perturbations at high throughput — a capability highly relevant for regenerative applications and quality assessment before cell transplantation.
Demonstrated in peer-reviewed research:
High-throughput mechanophenotyping has been used to quantify mechanical maturation and heterogeneity in cardiomyocyte populations under defined differentiation conditions. The authors applied real-time deformability cytometry to hiPSC-CMs, quantifying elasticity and deformation across thousands of cells, and linked mechanical transitions to cytoskeletal contributions.